Ionic character11/12/2023  
Molecular polarity : A molecule is polar if it has a net dipole moment, which depends on the existence of polar covalent bonds and the molecule’s three-dimensional structure or geometry.ĭipole: Usually refers to the separation of charges within a molecule between two covalently bonded atoms or atoms that share an ionic bond It is the product of the charge on either pole of the dipole and the distance separating them. Dipole moments can be used to measure the strength of the dipole by multiplying the separated charge and bond length.Įlectronegativity: The tendency of an atom or molecule to attract electrons and form bonds.īond polarity: A covalent bond is polar if one atom is more electronegative than its bonding partner, resulting in a net dipole moment between the two atoms.ĭipole moment: A measure of the polarity of a covalent bond or an entire molecule.Polarity can be in a bond when the two elements have a different electronegativity or in a molecule when one atom draws the majority of the electron density creating an area of negative charge.Bonds which are not purely ionic or covalent due to differences in electronegativity are polar molecules.Bonding between elements can be viewed on a spectrum of ionic to covalent character.The dipole moment is calculated by evaluating the product of the magnitude of separated charge, q, and the bond length, r. The strength of a dipole’s interaction with an electric field is given by the electric dipole moment of the bond or molecule. A polar molecule acts as an electric dipole which can interact with electric fields that are created artificially, or that arise from interactions with nearby ions or other polar molecules.ĭipoles are conventionally represented as arrows pointing in the direction of the negative end. 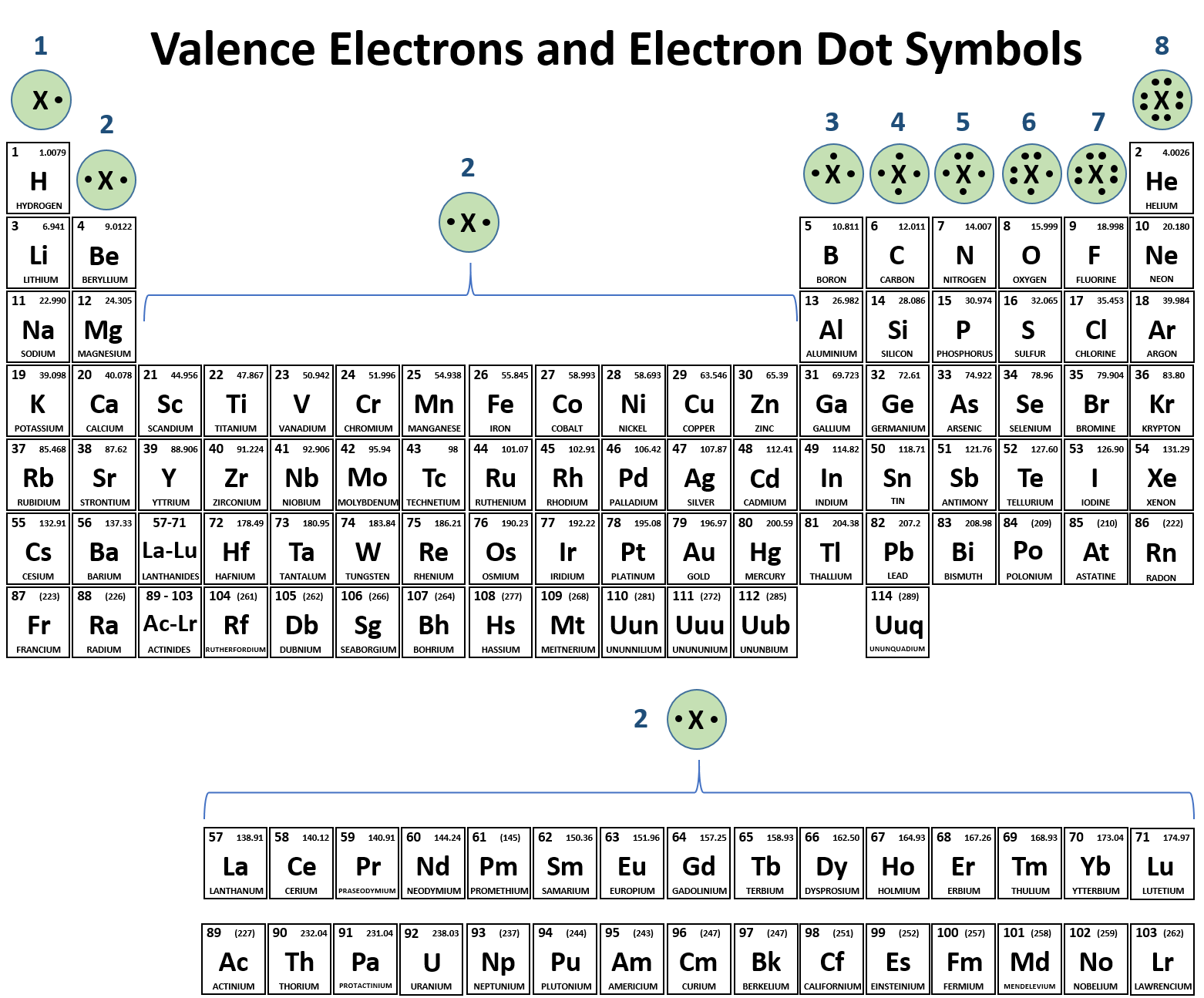
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
